When did dalton form his atomic theory2/18/2024  
Whatever names it may go by, they still signify one and the same power. 
These observations have tacitly led to the conclusion which seems universally adopted, that all bodies of sensible magnitude, whether liquid or solid, are constituted of a vast number of extremely small particles, or atoms of matter bound together by a force of attraction, which is more or less powerful according to circumstances, and which as it endeavours to prevent their separation, is very properly called in that view, attraction of cohesion but as it collects them from a dispersed state (as from steam into water) it is called, attraction of aggregation, or more simply affinity. In steam we recognise a perfectly elastic fluid, in water a perfect liquid, and in ice a complete solid. A very famous instance is exhibited to us in water, of a body, which, in certain circumstances, is capable of assuming all the three states. There are three distinctions in the kinds of bodies, or three states, which have more especially claimed the attention of philosophical chemists namely, those which are marked by the terms elastic fluid, liquids, and solids. But before this subject can well be considered, we should first settle what is intended to be meant by the word temperature. It is very certain that the two methods will not give precisely the same results, because the expansions of different bodies by equal increments of temperature are not the same. From the experiments hitherto made there seems little doubt of its being nearly so but it is perhaps more correct to deduce the specific heat of bodies from equal bulks than from equal weights. Whether the specific heats, could they be thus obtained for one temperature, would express the relation at every other temperature, whilst the bodies retained their form, is an enquiry of some moment. Attempts of this kind have been made with very considerable success. Were the whole quantities of heat in bodies of equal weight or bulk, or even the relative quantities, accurately ascertained, for any temperature, the numbers expressing those quantities would constitute a table of specific heats, analogous to a table of specific gravities, and would be an important acquisition to science. Each kind of matter has its peculiar affinity for heat, by which it requires a certain portion of the fluid, in order to be in equilibrium with other bodies at a certain temperature. When all surrounding bodies are of one temperature, then the heat attached to them is in a quiescent state the absolute quantities of heat in any two bodies in this case are not equal, whether we take the bodies of equal weights or of equal bulks. The most probable opinion concerning the nature of caloric, is, that of its being an elastic fluid of great subtility, the particles of which repel one another, but are attracted by all other bodies. Thomas Thomson's account of Dalton's atomism also preceded the publication of New System of Chemical Philosophy. A "Table of the relative weights of the ultimate particles of gaseous and other bodies" appears in Dalton 1805b. It is not, however, the first published statement of Dalton's atomic hypothesis or the first table of his atomic weights. It also includes some of Dalton's ideas on heat and gases, ideas which were later to hinder the acceptance of Avogadro's hypothesis ( Chapter 9). The selection reproduced in this chapter includes Dalton's atomic hypothesis as he enunciated it in his New System of Chemical Philosophy. The law of multiple proportions came to be regarded as an empirical law quite independent of its relation to the atomic hypothesis or perhaps as an empirical law that inspired the atomic hypothesis however, Roscoe and Harden have shown that in Dalton's mind it was a testable prediction which followed from the atomic hypothesis. The law of definite proportions or of constant composition had previously been proposed in the work of Jeremias Richter and Joseph-Louis Proust. The laws of definite and multiple proportions are also associated with Dalton, for they can be explained by his atomic hypothesis. Dalton is also associated with color blindness, sometimes called daltonism, a condition from which he suffered and which he was the first to describe. His experiments on gases also led to a result now known as Dalton's law of partial pressures. 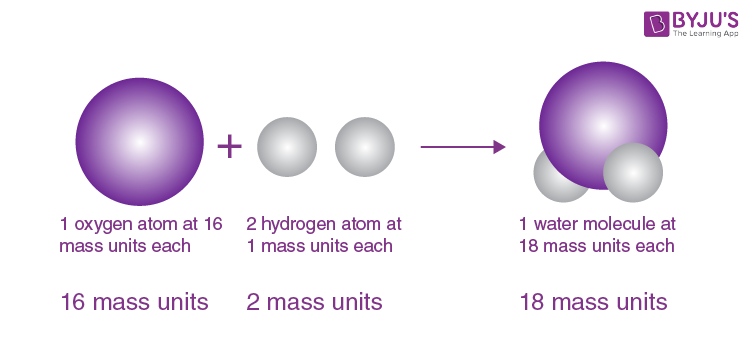 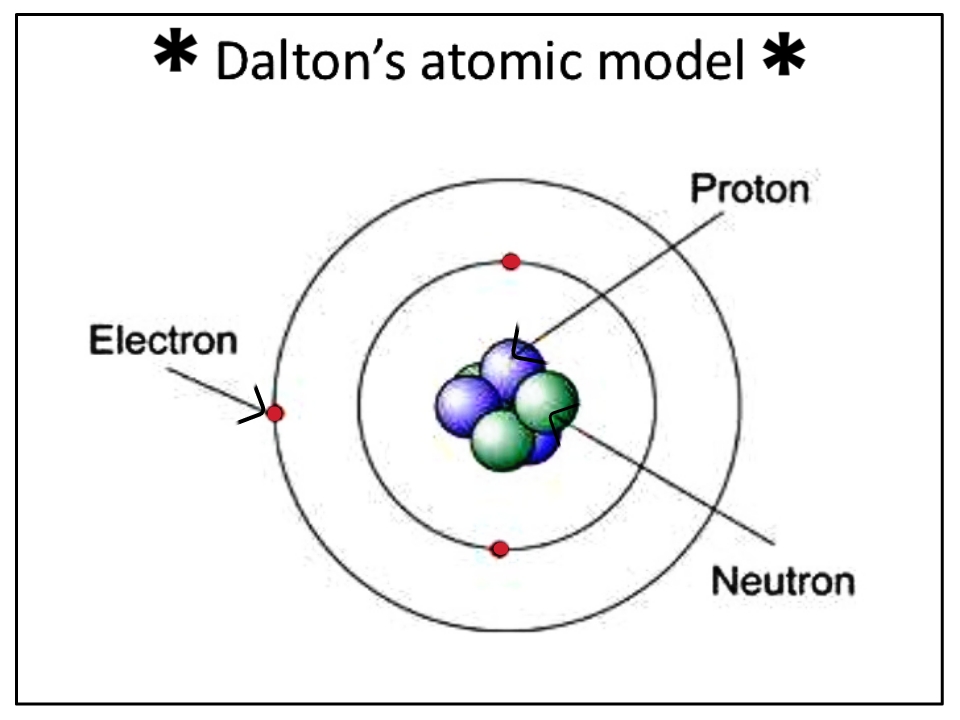
Indeed, it was this interest which led him to study gases, out of which study grew his atomic hypothesis. Dalton had a life-long interest in the earth's atmosphere. John Dalton (1766-1844 view portrait at the National Portrait Gallery, London) was an English scientist and teacher, best known for turning the ancient notion of atoms into a scientific hypothesis with testable consequences. Atoms of Definite Weight: Dalton Elements and Atoms: Chapter 7 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
